The ROCKstar (KD025-213) study
Clinically meaningfula responses with REZUROCK were seen across all patient types after failure of ≥2 prior lines of therapy1,2

Clinically meaningfula and statistically significantb ORRc in a real-world demographicd of patients1,2

Clinically meaningfula ORRs observed across key subgroups2

Responses across all evaluated organs, including those with fibrotic manifestations2

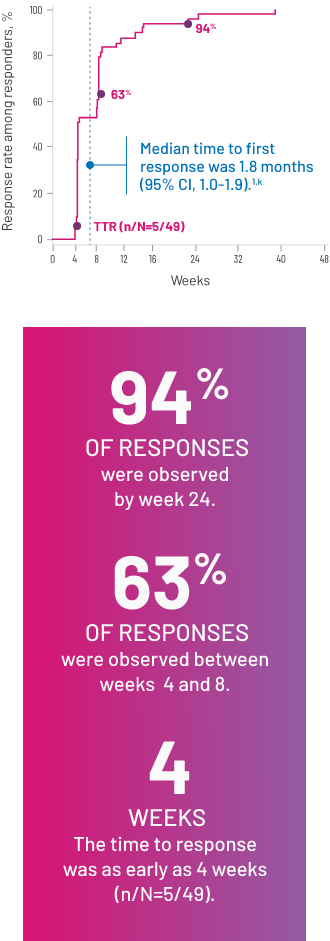
Most responses were seen between 4 and 8 weeks3

FFSe rate at 6 and 12 months and OSf rate at 24 months2,3

Exploratory analyses of LSSg summary score showed clinically meaningful improvements (≥7-point reduction) in patient-reported QOL1

Reduction in use of CS and CNI therapies2,3,h
aClinically meaningful was defined as a ≥30% ORR.2
bStatistical significance was achieved if the lower bound of the 95% CI of ORR exceeded 30%.2
cProportion of patients who achieved CR or PR according to the 2014 NIH cGVHD Consensus Criteria.1
dROCKstar study select baseline patient characteristics (200-mg once-daily arm): median age of 53 years (range, 21-77); male, n=42 (64%); median of 3 prior lines of systemic therapy; median of 25 months (range, 2-162) from cGVHD diagnosis to enrollment; median prednisone-equivalent dose at enrollment of 0.20 mg/kg/d (range, 0.03-0.95); concomitant PPI use, n=33 (50%); ≥4 organs involved, n=33 (50%); previous aGVHD, n=42 (64%); refractory to prior line of systemic therapy, n=44 (79%)i; NIH-defined disease severity: n=46 (70%) severe, n=18 (27%) moderate, n=2 (3%) mild. Prior systemic therapies included corticosteroids (prednisone), n=65 (99%); tacrolimus, n=40 (61%); ECP, n=31 (47%); ibrutinib, n=22 (33%); and ruxolitinib, n=20 (30%).2,3
eFFS was defined as the absence of relapse, nonrelapse mortality or a need for additional systemic therapy.2
fOS was defined as the time from the first dose of REZUROCK to the date of death due to any cause.3
gThe LSS is a 30-item, 7-subscale symptom scale and QOL measurement tool that evaluates the AEs of cGVHD in the categories of skin, vitality, lung, nutritional status, psychological functioning, eye and mouth.4
hThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
iDenominator excludes patients with unknown status.1
STATISTICALLY SIGNIFICANT ORRc FOLLOWING TREATMENT WITH REZUROCK 200 mg ONCE DAILY1,2,5
CR, n=4 (6%). PR, n=45 (69%).1
bStatistical significance was achieved if the lower bound of the 95% CI of ORR exceeded 30%.2
cProportion of patients who achieved CR or PR according to the 2014 NIH cGVHD Consensus Criteria.1
jBased on a final analysis by the FDA as seen through cycle 7 day 1 (n=65).1
CLINICALLY MEANINGFULa ORRs OBSERVED ACROSS KEY SUBGROUPS IN THE 200-mg ONCE-DAILY ARM3





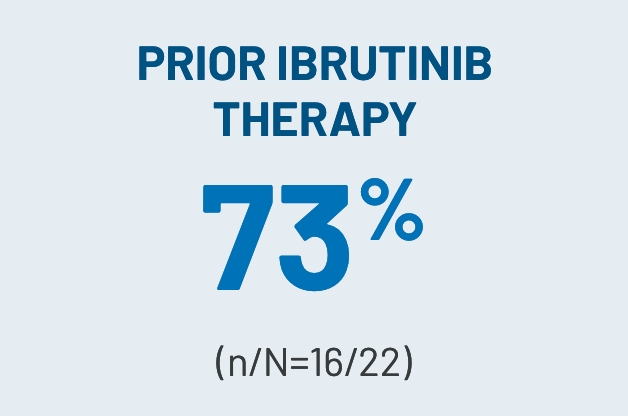
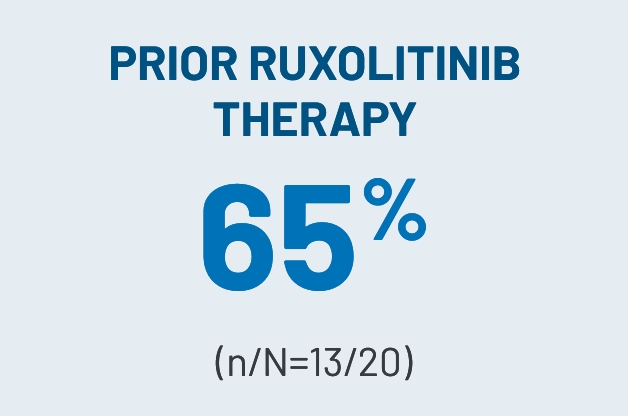
aClinically meaningful was defined as a ≥30% ORR.2
RESPONSES BY ORGAN SYSTEM WITH REZUROCK 200 mg ONCE DAILY IN THE mITT POPULATION (n=66)3
![Bar graph of complete and partial responses (CR and PR) by organ system with REZUROCK 200 mg once daily in the modified intent-to-treat (mITT) population. Upper GI tract [(n/N=8/13)(total response 62% with PR 8% and CR 54%)], esophagus [(n/N=9/19)(CR 47%)], lower GI tract [(n/N=4/6)(CR 67%)], mouth [(n/N=16/30)(total response 53% with PR 3% and CR 50%)], joints/fascia [(n/N=38/51)(total response 75% with PR 55% and CR 20%)], liver [(3/9)(total response 33% with PR 11% and CR 22%)], skin [(18/55)(total response 33% with PR 18% and CR 15%)], eyes [(16/48)(total response 33% with PR 16% and CR 17%)] and lungs [(n/N=7/24)(total response 29% with PR 12% and CR 17%)]](/dam/jcr:5644150a-5bf8-4f83-b5aa-344b439fc712/m-_2.1_bar_chart_outlined.png)
CR was observed in all evaluated organs, including those with fibrotic manifestations, such as the lungs, skin, joints/fascia and GI system.2
Most responses were seen between 4 and 8 weeks3
CUMULATIVE RESPONSE RATES OVER TIME IN THE RESPONDER POPULATION WITH REZUROCK 200 mg ONCE DAILY (n/N=49/66)3

did not experience death or new systemic therapy initiation (95% CI, 46-74) for ≥12 months after response.1
kBased on a final analysis by the FDA (n=65).
FFSe,l WITH REZUROCK 200 mg ONCE DAILY IN THE mITT POPULATION2,3
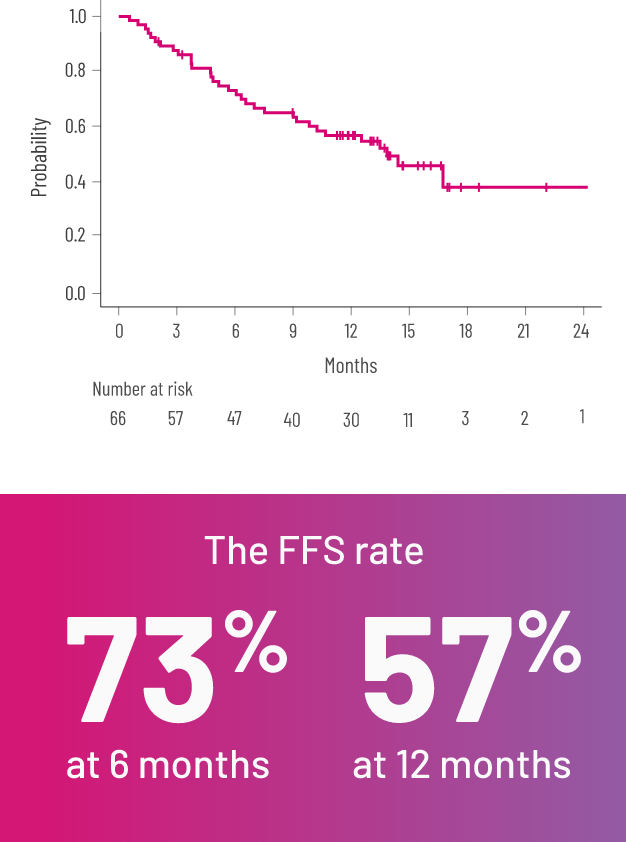
Kaplan-Meier curve of estimated FFS.
eFFS was defined as the absence of relapse, nonrelapse mortality or a need for additional systemic therapy.2
lPrespecified secondary end point; not powered to show statistical significance.
OSf,l WITH REZUROCK 200 mg ONCE DAILY IN THE mITT POPULATION3
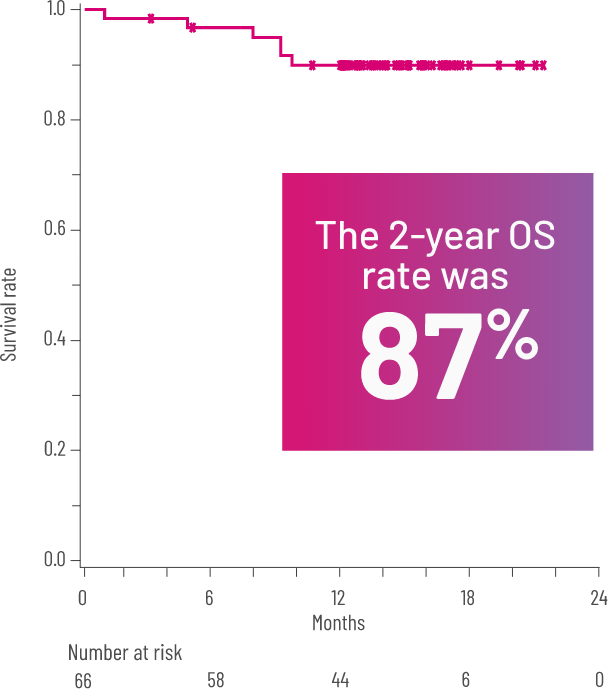
Kaplan-Meier curve of estimated OS.
fOS was defined as the time from the first dose of REZUROCK to the date of death due to any cause.3
lPrespecified secondary end point; not powered to show statistical significance.
CLINICALLY MEANINGFUL IMPROVEMENTS IN PATIENT-REPORTED QOL1,m
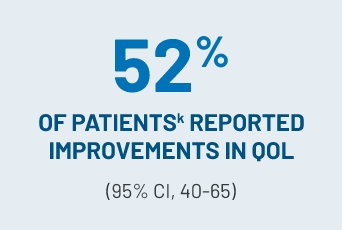
(≥7-point reduction in LSSg summary score) with REZUROCK 200 mg once daily in the mITT population in an exploratory analysis
- Both responders (61%) and nonrespondersn (25%) had improved QOL scores6
gThe LSS is a 30-item, 7-subscale symptom scale and QOL measurement tool that evaluates the AEs of cGVHD in the categories of skin, vitality, lung, nutritional status, psychological functioning, eye and mouth.4
kBased on a final analysis by the FDA (n=65).
mThrough cycle 7 day 1.1
nNonresponders were defined as patients with CR or PR in ≥1 organ, accompanied by progression in another organ (considered progression); outcomes that did not meet the criteria for CR, PR, progression or mixed response; or progression in ≥1 organ or site without a response in any organ or site.30
REDUCTION IN USE OF CS AND CNI THERAPIES IN THE 200-mg ONCE-DAILY ARM2,3,l
Patients who received CS therapy2,h



Patients who received CNI therapy3,h

hThe final FDA interpretation of the ROCKstar study omitted 1 patient from the REZUROCK 200-mg once-daily arm. As a result, there are minor differences between the ROCKstar publication, where n=66, and the Prescribing Information, where n=65.
lPrespecified secondary end point; not powered to show statistical significance.
nNonresponders were defined as patients with CR or PR in ≥1 organ, accompanied by progression in another organ (considered progression); outcomes that did not meet the criteria for CR, PR, progression or mixed response; or progression in ≥1 organ or site without a response in any other organ or site.7
TOLERABILITY IS KEY.8
AE, adverse event; aGVHD, acute graft-versus-host disease; cGVHD, chronic graft-versus-host disease; CNI, calcineurin inhibitor; CR, complete response; CS, corticosteroid; DOR, duration of response; ECP, extracorporeal photopheresis; FDA, US Food and Drug Administration; FFS, failure-free survival; GI, gastrointestinal; LSS, Lee Symptom Scale; mITT, modified intent-to-treat; MOA, mechanism of action; NIH, National Institutes of Healh; ORR, overall response rate; OS, overall survival; PPI, proton pump inhibitor; PR, partial response; QOL, quality of life.
References: 1. REZUROCK. Package insert. Kadmon Pharmaceuticals, LLC. 2. Cutler C, Lee SJ, Arai S, et al; on behalf of the ROCKstar Study Investigators. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138(22):2278-2289. doi:10.1182/blood.2021012021 3. Data on file 1. Kadmon Pharmaceuticals, LLC; 2021. 4. Lee SJ, Cook EF, Soiffer R, et al. Development and validation of a scale to measure symptoms of chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2002;8(8):444-452. doi:10.1053/bbmt.2002.v8.pm12234170 5. Data on file 2. Kadmon Pharmaceuticals, LLC; 2021. 6. Data on file 3. Kadmon Pharmaceuticals, LLC; 2021. 7. Data on file 4. Kadmon Pharmaceuticals, LLC; 2019. 8. Broadening the definition of tolerability in cancer clinical trials to better measure the patient experience. Friends of Cancer Research. Published October 24, 2018. Accessed February 26, 2024. https://www.focr.org/sites/default/files/Comparative Tolerability Whitepaper_FINAL.pdf
INDICATION
IMPORTANT SAFETY INFORMATION
IMPORTANT SAFETY INFORMATION
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
IMPORTANT SAFETY INFORMATION
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
IMPORTANT SAFETY INFORMATION
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
- Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, REZUROCK can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with REZUROCK and for one week after the last dose
Adverse Reactions
- The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension
- Permanent discontinuation of REZUROCK due to adverse reactions occurred in 18% of patients. The adverse reactions which resulted in permanent discontinuation of REZUROCK in > 3% of patients included nausea (4%). Adverse reactions leading to dose interruption occurred in 29% of patients. The adverse reactions leading to dose interruption in ≥ 2% were infections (11%), diarrhea (4%), and asthenia, dyspnea, hemorrhage, hypotension, liver function test abnormal, nausea, pyrexia, edema, and renal failure with (2% each)
- Monitor total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) at least monthly
Drug Interactions
- Strong CYP3A Inducers: Coadministration of REZUROCK with strong CYP3A inducers decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with strong CYP3A inducers
- Proton Pump Inhibitors: Coadministration of REZUROCK with proton pump inhibitors decreases belumosudil exposure, which may reduce the efficacy of REZUROCK. Increase the dosage of REZUROCK to 200 mg twice daily when coadministered with proton pump inhibitors
- Certain UGT1A1 substrates: Avoid coadministration of REZUROCK with UGT1A1 substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the UGT1A1 substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of UGT1A1. Coadministration of REZUROCK with a UGT1A1 substrate decreased plasma concentrations of the glucuronide metabolite, which may increase the risk of adverse reactions related to sensitive substrates of UGT1A1
- Certain P-gp, OATP1B1, and BCRP substrates: Avoid coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates, for which minimal concentration changes may lead to serious toxicities. If coadministration cannot be avoided, decrease the P-gp, OATP1B1, and BCRP substrates dosage(s) in accordance with the respective Prescribing Information. REZUROCK is an inhibitor of P-gp, OATP1B1, and BCRP. Coadministration of REZUROCK with P-gp, OATP1B1, and BCRP substrates increased their plasma concentrations, which may increase the risk of adverse reactions related to these substrates
Use in Specific Populations
- Pregnancy: There are no available human data on REZUROCK use in pregnant women to evaluate for a drug-associated risk. Advise pregnant women and females of reproductive potential of the potential risk to the fetus
- Lactation: There are no data available on the presence of belumosudil or its metabolites in human milk or the effects on the breastfed child, or milk production. Because of the potential for serious adverse reactions from belumosudil in the breastfed child, advise lactating women not to breastfeed during treatment with REZUROCK and for one week after the last dose
- Pediatric Use: The safety and effectiveness of REZUROCK in pediatric patients less than 12 years old have not been established
- Geriatric Use: Of the 186 patients with chronic GVHD in clinical studies of REZUROCK, 26% were 65 years and older. No clinically meaningful differences in safety or effectiveness of REZUROCK were observed in comparison to younger patients
- Renal Impairment: Treatment with REZUROCK has not been studied in patients with pre-existing severe renal impairment. For patients with pre-existing severe renal impairment, consider the risks and potential benefits before initiating treatment with REZUROCK
- Hepatic Impairment: Avoid use in patients with moderate hepatic impairment (Child-Pugh B) or severe hepatic impairment (Child-Pugh C) without liver GVHD. No dose adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A)
Please click here for full Prescribing Information.
INDICATION
REZUROCK® (belumosudil) is indicated for the treatment of adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
